Electrolysis Technologies
Advanced electrochemical technologies for hydrogen and sustainable fuel production
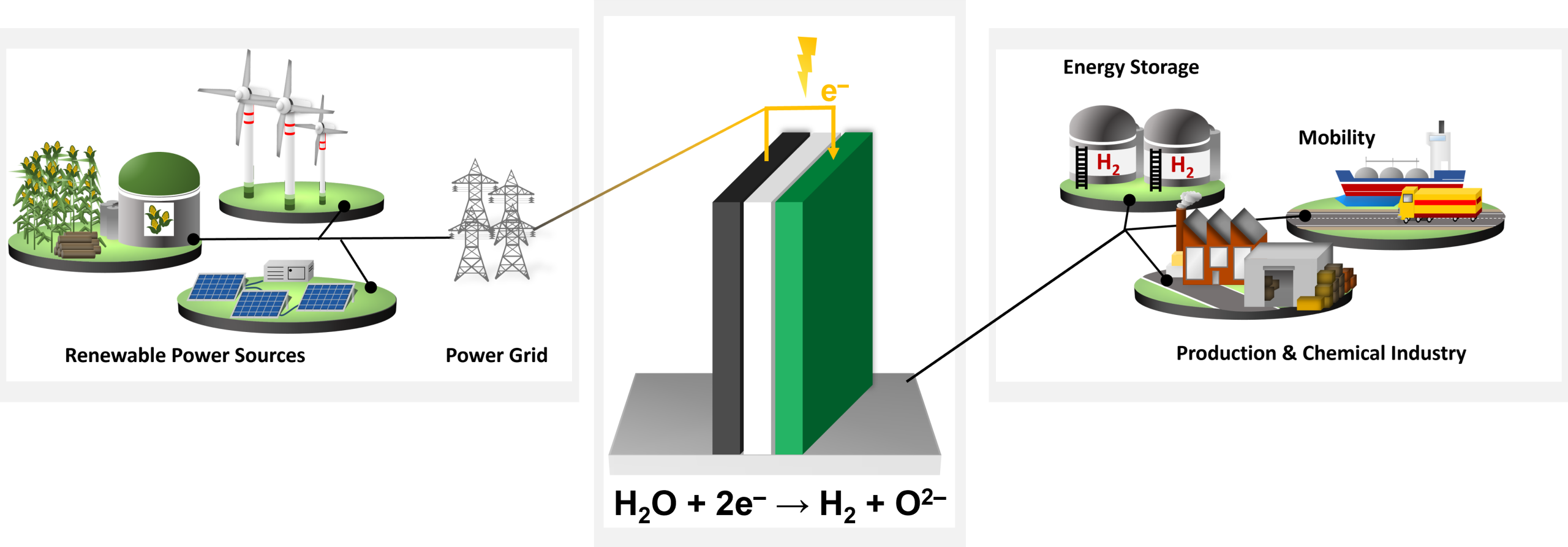
What Is Electrolysis?
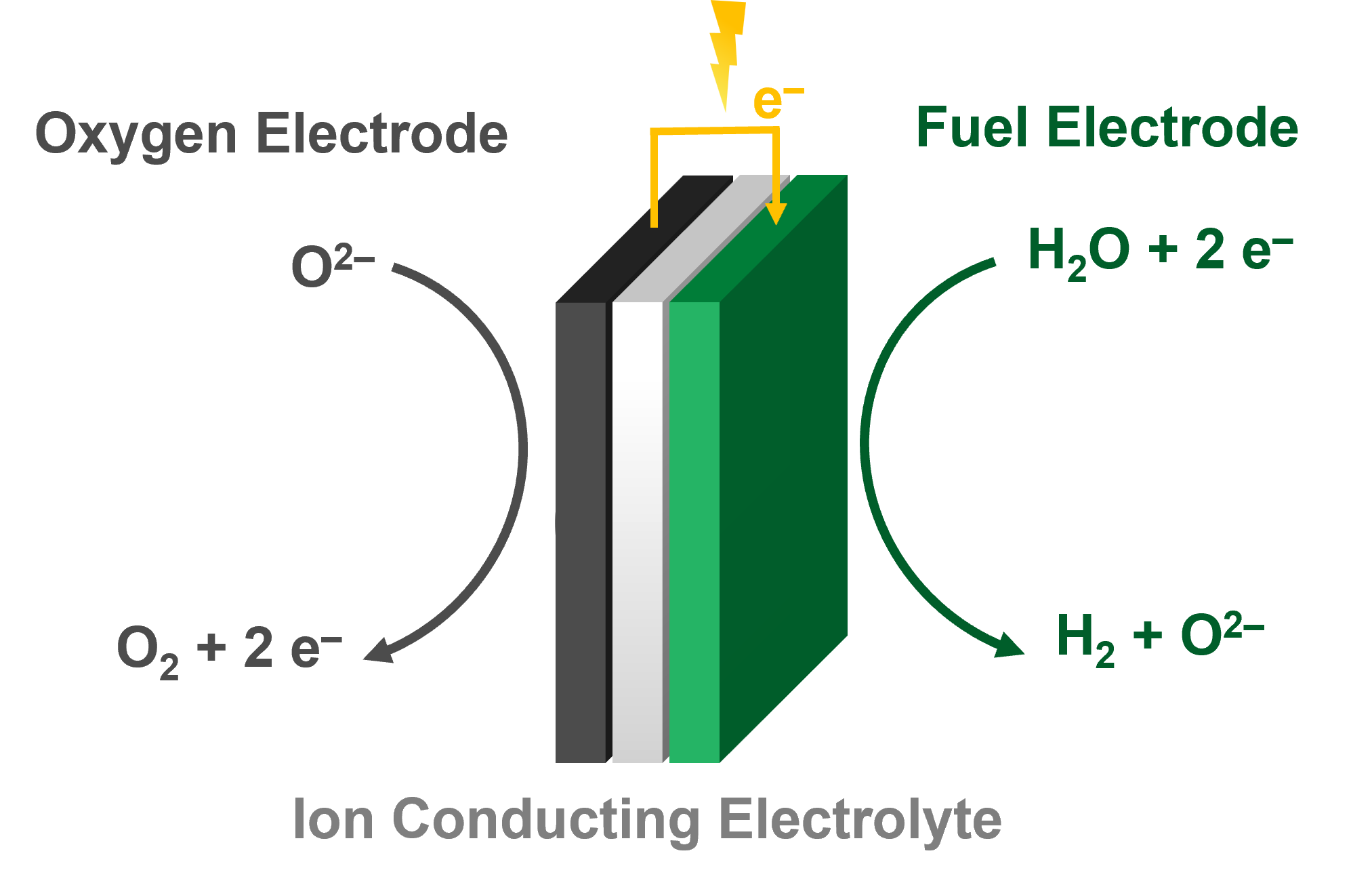
Electrolysis is the electrochemical process that converts electrical energy into chemical energy by splitting water into hydrogen and oxygen. When an external voltage is applied, water molecules at the cathode are reduced to form hydrogen gas, while water oxidation at the anode releases oxygen. The efficiency of this process depends on the ionic conductivity of the electrolyte, the catalytic activity of the electrodes, and the ability of the system to manage heat and mass transport.
In the context of a transition to sustainable energy systems, electrolysis enables the direct storage of renewable electricity in the form of hydrogen — a versatile energy carrier that can be stored, transported, and later reconverted into electricity or used as a feedstock for chemical synthesis. Hydrogen produced through electrolysis can decarbonize sectors such as steel manufacturing, refining, and chemical synthesis, and can serve as a building block for power-to-X pathways (e.g., synthetic fuels, ammonia).
Electrochemical impedance spectroscopy (EIS) plays a critical role in understanding electrolysis performance and degradation mechanisms. By probing the frequency-dependent response of an electrochemical cell, EIS separates resistive and capacitive contributions arising from bulk ionic transport, interfacial charge transfer, and electrode processes — making it indispensable for optimizing materials and cell designs.
Electrolysis Technology Platforms
Electrolyzers vary in component materials and operating conditions, each optimized for specific conditions of temperature, reactants, and targeted application options.
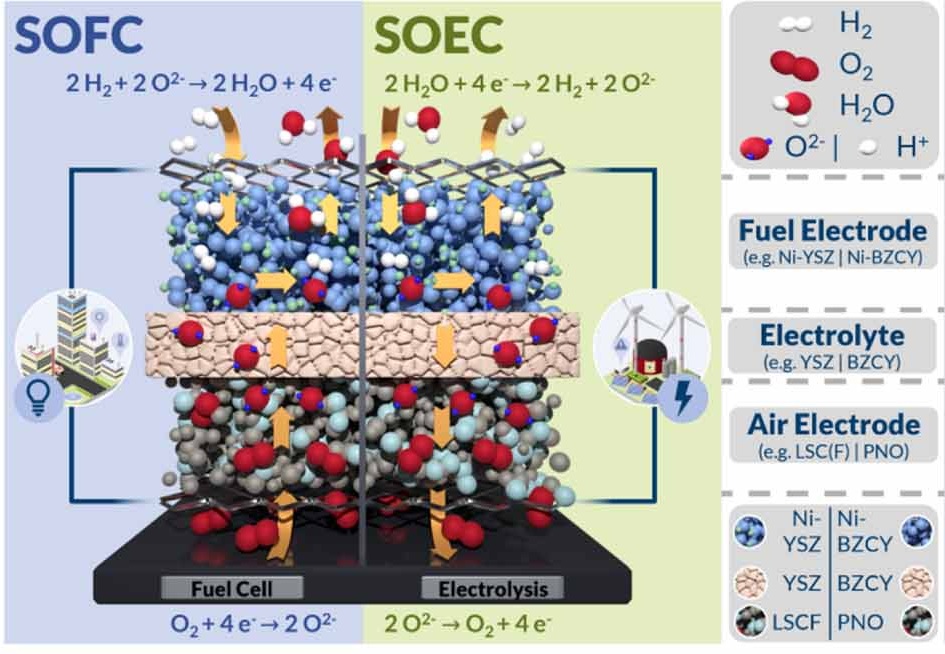
Solid Oxide Electrolysis (SOEC)
High-temperature operation
High electrical efficiency
co-electrolysis (H₂O + CO₂ → syngas)

Alkaline Electrolysis (AEL)
Mature, cost-effective
Liquid electrolyte
Large-scale hydrogen production

Proton Exchange Membrane Electrolysis (PEMEL)
Polymer electrolyte
High current density
Dynamic operation with renewables

Proton-Conducting Ceramic Electrolysis (PCEC)
Intermediate temperature
Ceramic proton-conducting electrolytes
Reduced degradation potential

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Materials & Components — Cell Level
The performance and durability of electrolysis technologies are governed not only by operating conditions but fundamentally by the materials and components used within the cell and stack. Transport properties, interfacial stability, and mechanical compatibility between components strongly influence efficiency and degradation behavior. While these fundamental components are common to all electrolysis technologies, their material requirements and operating challenges vary significantly with temperature. In high-temperature electrolysis systems, component design must additionally account for enhanced transport rates, thermochemical effects, and long-term stability under extreme operating conditions.
Electrolytes
For high-temperature electrolysis, ceramic electrolytes like yttria-stabilized zirconia (YSZ) or gadolinium-doped ceria (GDC) are widely used. These materials exhibit high oxide-ion conductivity at elevated temperatures and enable efficient ion transport between electrodes. Current research focuses on improving ionic conductivity while minimizing degradation mechanisms such as grain growth and chemical instability at interfaces.
Oxygen Electrodes (Anodes)
The oxygen electrode must catalyze the oxygen evolution reaction (OER) while maintaining structural integrity under oxidizing conditions at high temperatures. Conventional perovskite materials such as LSC(F) show good performance but suffer from strontium segregation and secondary phase formation at interfaces, which degrade long-term performance. Emerging electrode materials, including layered Ruddlesden–Popper nickelates and double perovskites, exhibit enhanced oxide ion diffusion and surface exchange activity, resulting in improved performance and potentially lower degradation rates.
Fuel Electrodes (Cathodes)
Nickel-based cermet electrodes such as Ni-YSZ and Ni-GDC are state-of-the-art for hydrogen electrodes. However, under SOEC conditions, nickel agglomeration and migration can alter microstructure and reduce active surface area over time. Alternative mixed ionic–electronic conducting materials are under investigation to enhance chemical stability and durability while maintaining adequate catalytic activity.
Materials & Components — Stack & System Level
Beyond individual cells, the full electrolyzer stack incorporates additional engineering components that govern scale-up, thermal management, and system integration.
Interconnects & Bipolar Plate
These components provide electrical pathways between cells and separate gas streams. Material choices must balance electrical conductivity, oxidation resistance at high temperature, and coefficient of thermal expansion compatibility with adjacent cell materials.
Seals & Coatings
Gas-tight seals prevent cross-contamination of reactants in the stack and must endure long-term thermal cycling without cracking or creeping. Protective coatings on metallic interconnects and cell surfaces can mitigate corrosion and extend service life.
Thermal & Flow Management Systems
Efficient heat distribution and reactant supply are vital for uniform cell operation. For SOEC systems, thermal integration with external heat sources and waste heat recovery are major design considerations that can significantly influence overall system efficiency.
